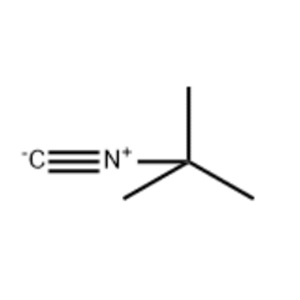
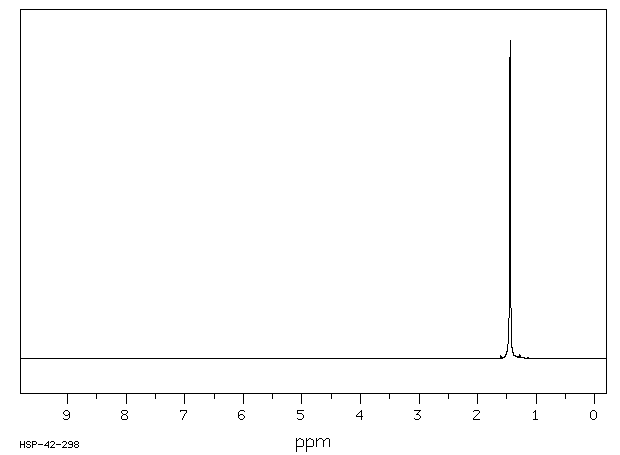
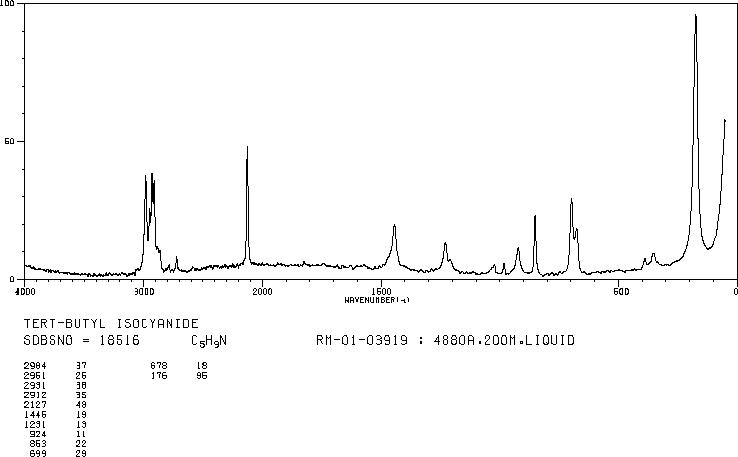
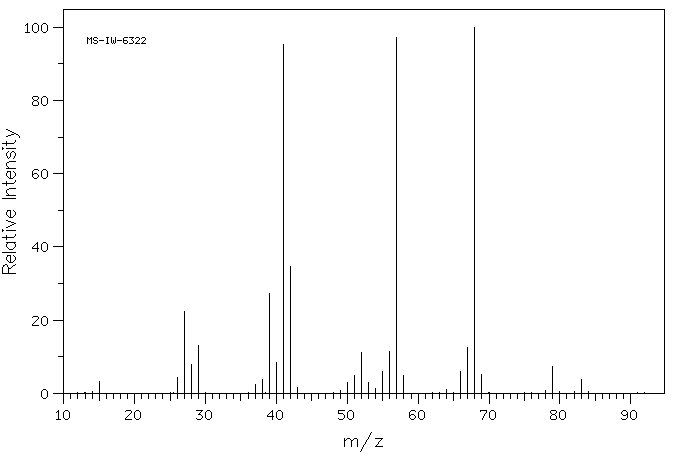
Alcoholics Anonymous Alcoholism Pie chart Al-Anon/Alateen, central business district, text, logo, pie png | PNGWing
![Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/33325e32-cff5-492a-88d0-118a8d83da04/mcontent.jpg)
Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library

PDF) Proc. of ACM CIKM 2008 Workshop on Improving Non-English Web Searching (iNEWS08) | Jesus Vilares - Academia.edu
![Type II intramolecular [5+2] cycloaddition: facile synthesis of highly functionalized bridged ring systems. | Semantic Scholar Type II intramolecular [5+2] cycloaddition: facile synthesis of highly functionalized bridged ring systems. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/49884cf03ca69fd94669bbcc238f0c9e600f1978/1-Figure1-1.png)
Type II intramolecular [5+2] cycloaddition: facile synthesis of highly functionalized bridged ring systems. | Semantic Scholar
![Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/f2abf488-937d-492a-a883-d90f90a0cf60/msch002.jpg)
Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library
![Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/c0533e04-7e20-4a97-b8bb-9dcfd8e098e2/msch003.jpg)
Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library
![Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/56dc8efe-66c7-4590-a059-2434cbbd9fb4/mfig001.gif)
Type II Intramolecular [5+2] Cycloaddition: Facile Synthesis of Highly Functionalized Bridged Ring Systems - Mei - 2015 - Angewandte Chemie International Edition - Wiley Online Library
![Tunable Key [3 + 2] and [2 + 1] Cycloaddition of Enaminones and α‑Diazo Compounds for the Synthesis of Isomeric Isoxazoles: Metal-Controlled Selectivity - ScienceDirect Tunable Key [3 + 2] and [2 + 1] Cycloaddition of Enaminones and α‑Diazo Compounds for the Synthesis of Isomeric Isoxazoles: Metal-Controlled Selectivity - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S1523706023004741-ol3c00636_0007.jpg)