Molecules | Free Full-Text | Synthesis of Phosphorus(V)-Substituted Six-Membered N-Heterocycles: Recent Progress and Challenges

Palladium-Catalyzed Domino Heck/Sulfination: Synthesis of Sulfonylated Hetero- and Carbocyclic Scaffolds Using DABCO–Bis(sulfur dioxide) | Organic Letters

Scheme 1 A catalytic cycle for palladium(0)-mediated benzylphosphonate... | Download Scientific Diagram

Postfunctionalization of the main chain of Poly(3-hexylthiophene) via anodic C–H phosphonylation | Polymer Journal

PDF) The Palladium Acetate‐Catalyzed Microwave‐Assisted Hirao Reaction without an Added P‐Ligand as a “Green” Protocol; A Quantum Chemical Study on the Mechanism

Palladium-catalyzed dehydrogenative coupling of terminal alkynes with secondary phosphine oxides - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC09567G

Palladium-Catalyzed C–P Bond-Forming Reactions of Aryl Nonaflates Accelerated by Iodide | The Journal of Organic Chemistry

Merging Electrolysis and Nickel Catalysis in Redox Neutral Cross-Coupling Reactions: Experiment and Computation for Electrochemically Induced C–P and C–Se Bonds Formation | CCS Chem

Palladium-Catalyzed C–P Bond-Forming Reactions of Aryl Nonaflates Accelerated by Iodide - ScienceDirect
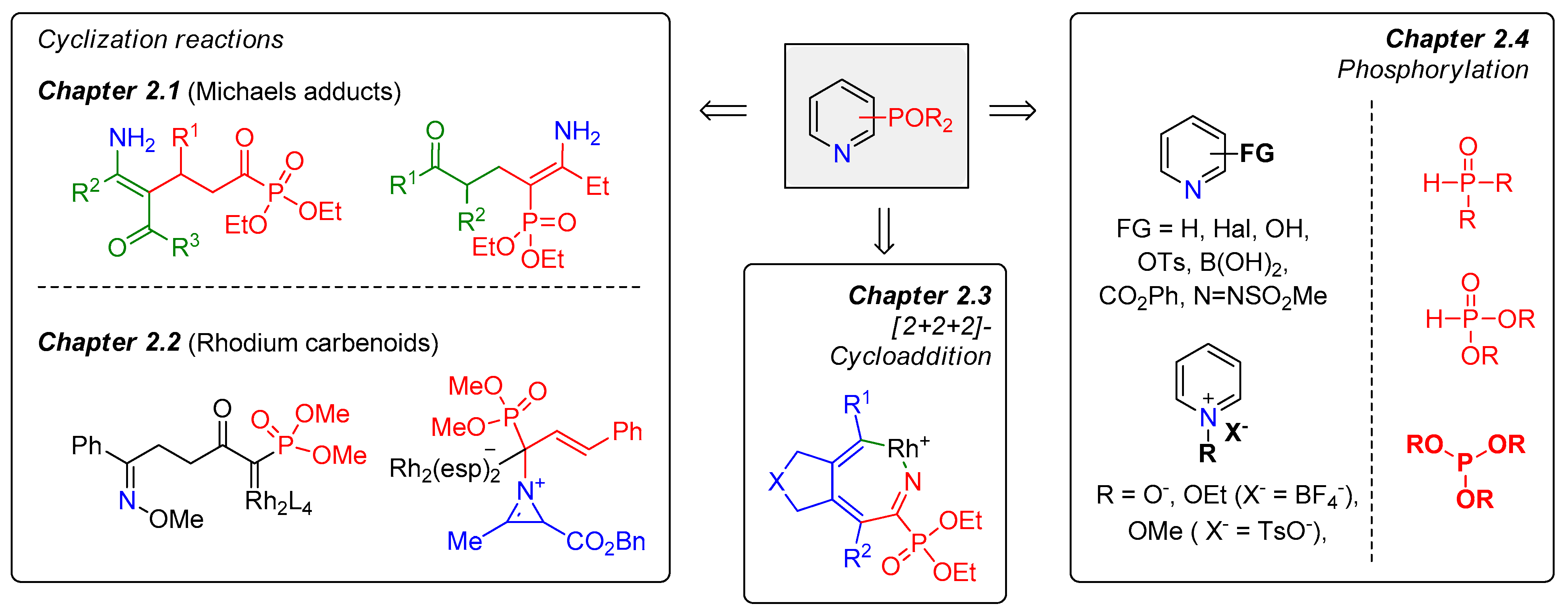
Molecules | Free Full-Text | Synthesis of Phosphorus(V)-Substituted Six-Membered N-Heterocycles: Recent Progress and Challenges
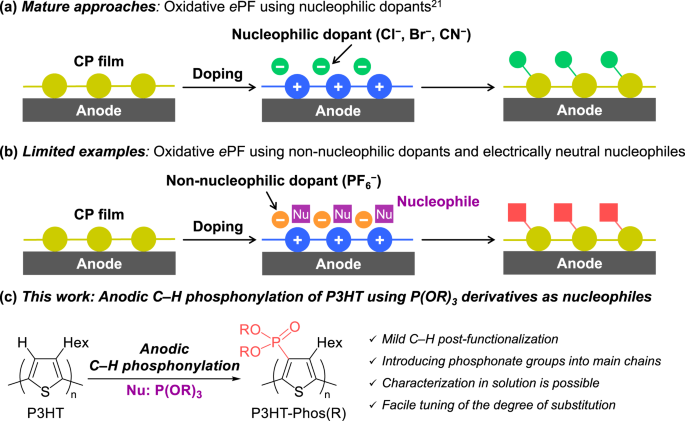
Postfunctionalization of the main chain of Poly(3-hexylthiophene) via anodic C–H phosphonylation | Polymer Journal
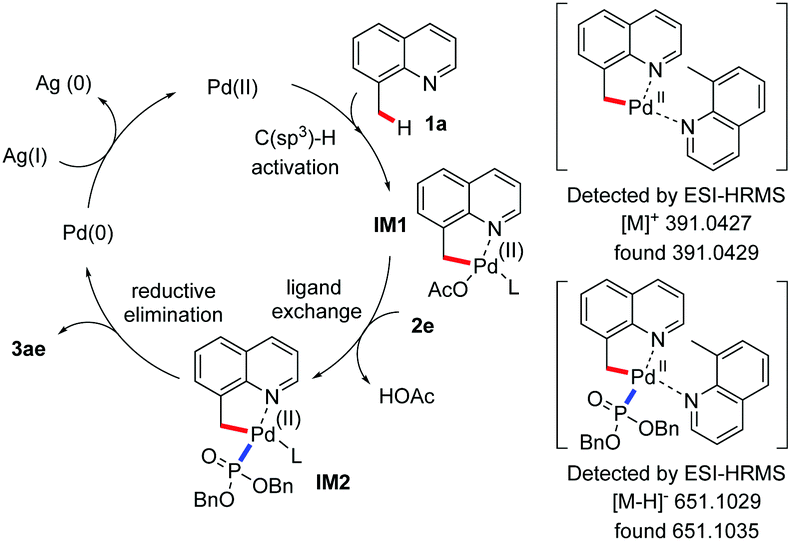
Palladium( ii )-catalyzed oxidative C(sp 3 )–P bond formation via C(sp 3 )–H bond activation - Chemical Communications (RSC Publishing) DOI:10.1039/C9CC07637A

Palladium‐Catalyzed Remote C−H Phosphonylation of Indoles at the C4 and C6 Positions by a Radical Approach - Shi - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Palladium-catalyzed desulfitative C–P coupling of arylsulfinate metal salts and H-phosphonates | Semantic Scholar

Palladium-Catalyzed C–P Bond-Forming Reactions of Aryl Nonaflates Accelerated by Iodide | The Journal of Organic Chemistry

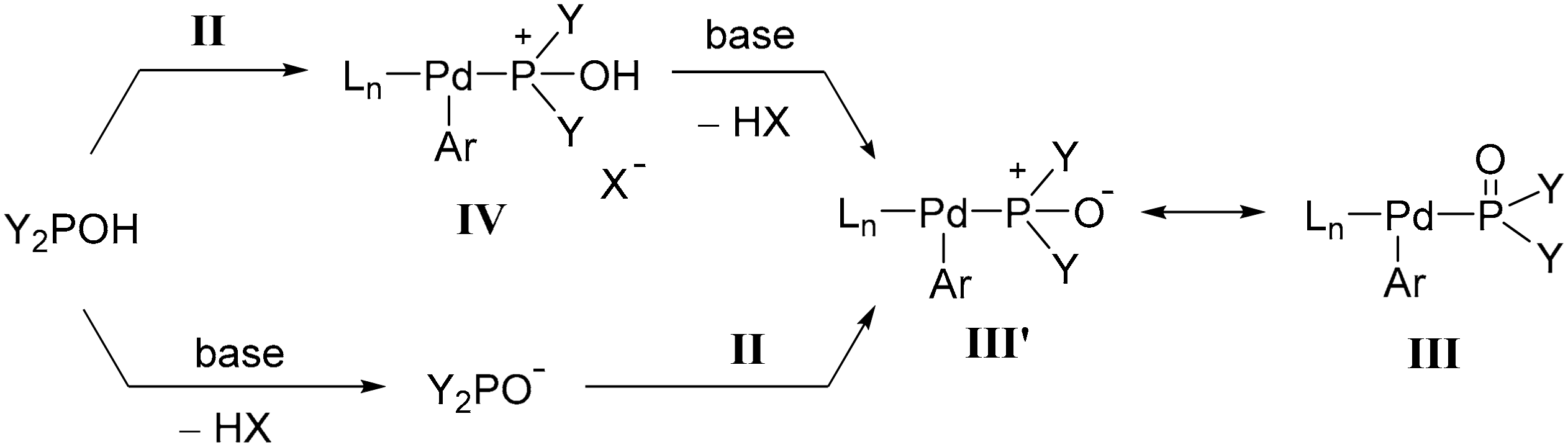
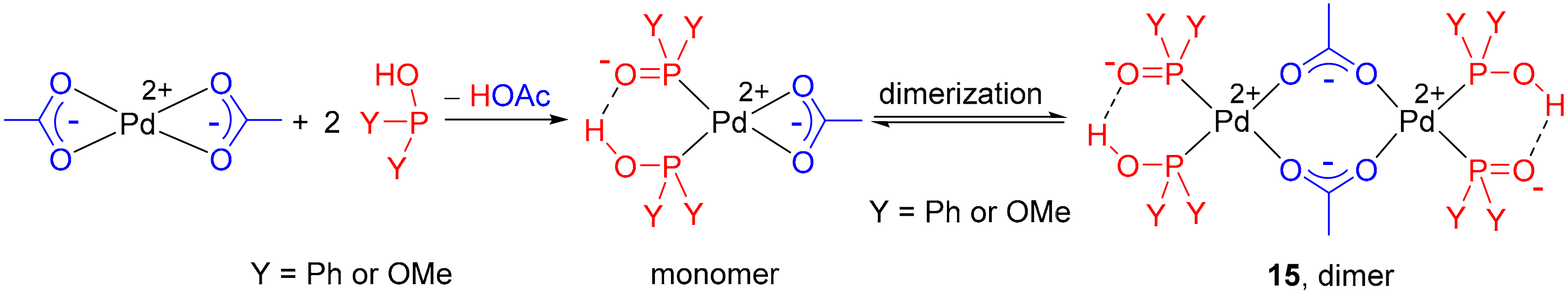
Palladium-catalyzed microwave-assisted Hirao reaction utilizing the excess of the diarylphosphine oxide reagent as the P-ligand; a study on the activity and formation of the “PdP2” catalyst

Phosphonylation of 2‐Amino‐ and 2‐Amido‐3‐bromopyridines and 2‐Amino‐3‐chloroquinoxalines with Triethyl Phosphite - Adam - 2009 - European Journal of Organic Chemistry - Wiley Online Library

Hydrophosphorylation of Alkynes Catalyzed by Palladium: Generality and Mechanism | Journal of the American Chemical Society

PDF) The Palladium Acetate‐Catalyzed Microwave‐Assisted Hirao Reaction without an Added P‐Ligand as a “Green” Protocol; A Quantum Chemical Study on the Mechanism